Capabilities
CTCReveal™ offers a non-invasive method for detecting cancer before imaging results appear in tissue biopsies. This is achieved through a blood-based, liquid biopsy, designed for individuals who want to proactively monitor their health. AI-assisted imaging offers more value for the client with enhanced data analysis.
A History of Proven Excellence
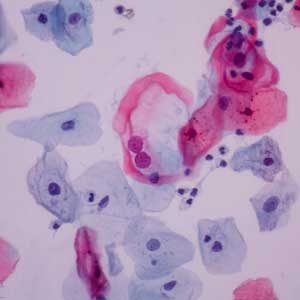
Danner Laboratory has operated for over 40 years as a CLIA-certified leader in molecular pathology. Placing a strong emphasis on our Quality Management System (QMS), we have processed over 100,000 specimens annually, with an emphasis on PAP smears and cervical cancer screening. Danner employs CAP certified cytologists that work diligently by our established high-quality systems. This has led to over 30,000 PAPs examined with a 99.5% satisfactory rate and greater than 95% biopsy concordance.
Studies have shown that regular PAP screening has a mortality reduction rate of at least 80%¹ with other studies showing up to 90%.

Early Detection for Better Recovery
CTCReveal™ enables individuals to get a head start on the cancer recovery process. This service reveals valuable information about biological risks before any tumors may appear visible. The process is safe and repeatable which enables proactive monitoring. Recognizing early warning signs before cancer becomes clinical allows for greatly improved odds of recovery.
CTCReveal™ allows patients to gain an understanding of their personal health risks, and preventive measures to help improve their conditions. We encourage our patients with individualized lifestyle adjustments to make based on their own scientific biomarkers, and livelihoods.
.
AI-Assisted Imaging Analysis
Artificial intelligence helps us to identify subtle CTC (circulating tumor cell) or CTM (circulating tumor microemboli) morphological patterns. This provides increased sensitivity for early-stage cancer changes.
AI assistance also supports a higher volume of early metastatic screening, while reducing human variability in cytopathology readings. Our AI training frameworks integrated with multi-source biological data supports a future for accurate, personalized predictions.

White Text
Current Applications
Utilizing CellBio™ Microfiltration, we are able to isolate CTCs and capture high quality samples, which results in high quality interpretation.

CellBio™ a2000
The GBC CellBio™ a2000 is an automated liquid biopsy platform that uses iFiltration™ technology to efficiently capture and harvest circulating tumor and other abnormal cells from blood for clinical and research applications.
Key Features
•Fast processing time
•95% recovery rate
•Single cell sensitivity
•Precise patented microfilter
CellSpot DT-300™
Once CTCs have been detected on a slide, we are able to scan images of them utilizing GBC’s CellSpot DT-300™ system. It is capable of a 40x zoom and storage of 300 slides at once, with automatic slide recognition.
Utilizing AI assistance, we are able to optimize quantitative analysis for cells and biomarkers. Analyses done on these CTCs provide great evidence for the future of preventive oncology and early metastatic cancer screening. After slides are scanned and analysis is conducted, instant accessibility of image data is acquired for sharing, education, or consultation with experts for research use.

How does it work?
CellBio™ Microfiltration
This 2025 Edison Award winning system captures intact CTCs and CTM by size and structural integrity. The microfilter membranes allow red blood cells to pass through, while preserving the natural morphology of the cancer cells. This provides better sensitivity for rare phenotypes and allows for a very detailed cytopathologist review.
AI Image Analysis
Artificial intelligence is used in the CellSpot DT-300 scanner to assist our cytopathologists and help optimize CTC detection. This AI assistance can recognize early-stage changes that might be undetectable by humans. This model also improves the consistency of rates in which CTCs and CTM are detected, while also tuning an algorithm for future detective patterns.

We receive a sample of blood from the patient.

The blood sample is prepared and microfiltered via CellBio™ a2000.

The sample is stained and AI-assisted imaging prepares final images.

Cytopathologist reviews results after cytologists analyze images. Results are returned to the patient.
White Text
Future Applications
.Cell Sequencing
Once CTCs are isolated from a blood sample, advanced genomic sequencing can be performed to analyze their DNA and RNA. This allows us to identify cancer-driving mutations, evaluate biological aggressiveness, and assess metastatic potential. By understanding the molecular profile of early cancer cells, we enable more precise monitoring, personalized therapy selection, and improved long-term outcomes.
Proteomic Analysis
Advanced proteomic analysis can be performed on isolated CTCs to evaluate the proteins driving tumor behavior. This can reveal which biological pathways are actively functioning such as growth signaling, metastatic potential, and early drug resistance mechanisms. These functional insights enable more precise risk stratification and support personalized therapeutic strategies before tumors are radiographically detectable.
Drug Development
Circulating tumor cell isolation provides direct access to the cells responsible for metastasis. Through genomic and proteomic characterization, novel therapeutic targets can be identified, enabling the development of anti-metastatic and prevision oncology drugs. Additionally, longitudinal CTC monitoring enhances clinical trial stratification, accelerates biomarker validation, and supports real-time assessment of therapeutic response.

white text


